Liquid conductivity and temperature sensor probe
52,00 € excluding VAT
Liquid conductivity and temperature probe.
Delivery within 2-3 days across the EU and 7-10 days worldwide
*Delivery dates may change based on your quantity and delivery address.
All orders are delivered by DHL ![]()
Enjoy free worldwide shipping on orders over 500EUR
Out of stock
Features
• Measure range 0 – 200 µS/cm
• Cell constant K = 0,1 or K=1
• Accuracy ± 0,5%
• Electrode materials stainless steel and ABS
• Pressure 2 bar
• Temperature up to 100 °C
• Temperature compensation Pt100
How to choose your conductivity probe to your application?
Conductivity is a measure of the ability of a fluid to conduct an electrical current. In water this ability is due to the presence of various ionized substances in solution. Conductivity measurements usually include temperature compensation.
First of all, you need to know the details of your project. Will the expected conductivity be high or low? Will the temperature change? In what area will it be used the probe? Based on these, you will be able to choose the one that best suits your purpose.
One of the most important characteristics, on the basis of which you should choose between sensors, is the cell constant or K factor. This number also describes the geometry of the probe. Based on this, it can be decided which liquids it is suitable for measuring.
If the cell constant is low, the distance between the conductor surfaces will be also small thus, the amount of liquid to be measured in the probe is also smaller. The small cell constant probes are used mostly in pure and ultrawater.
The larger the cell constant, the larger the sensing surfaces in the probe, the more liquid enters in it. These sensors are intended for use with liquids where the expected conductivity is higher than in the case of the pure water.
Based on the table below, we can determine the approximate cell constant probe we will need.
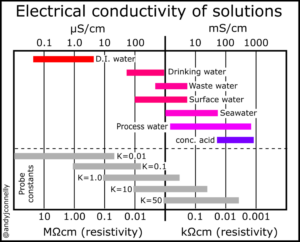
| Cell constant | 0.1, 1 |
|---|
Only logged in customers who have purchased this product may leave a review.
















Reviews
There are no reviews yet.